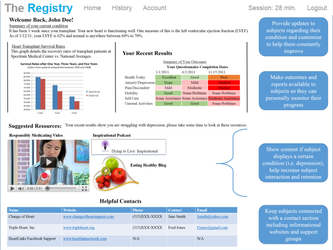
Overview
Imagine automatically building dynamic multi-media, logic driven content (e.g., charts, graphs, conditional text and formatting, etc.) based entirely on a Subject's OWN data! Learn more and see examples here.
Common Uses
| Clinical Trials | Patient Registries | Subject Portal |
|---|
- Serious Adverse Event and Adverse Event reporting
- Other event reports (e.g., protocol violation, randomization / enrollment, etc.)
- Invoice creation and reconciliation
| - Automated clinic notes
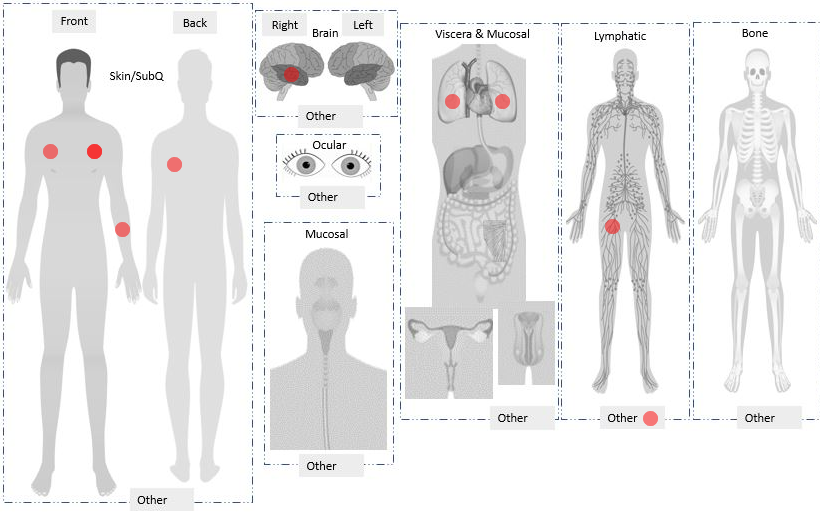
- Data visualization (e.g., charts, graphs, overlay images [i.e., plot tumor locations; see article here])
- Case conference notes (e.g., across specialties summaries [i.e., see Clinical Report design article here])
| - Dynamic newsletter based on preferences, milestones, etc.
- Outcome goals monitoring and points achievement
- Delivery of targeted educational materials (e.g., symptom triggered training materials, such as videos - website links - pods casts - etc.)
|
Examples:
| Reports for Subjects | Reports for Investigators |
|---|
|  |
| Conditional Formatting | Adverse Event |
|---|
| |
| Visit Summary | Automated Image Overlay (e.g., plot tumor locations) |
|---|
| |
| Invoice | Device Prescription |
|---|
| |
Video: Introduction To the New Subject Report Features
Watch a video that provides an overview of the new Subject Report features here.
New Features (Studytrax Version 3.47)
- Expression Builder
- Conditional Formatting
- The "Repeater" Control
- Charts
- Javascript editor
More Information
- Key Features
- Access to ALL data elements
- Integrated word-processor for layout design, formatting, etc.
- Design content based on a subject's individual needs using any web-based tool such as:
- Example content
- Charts
- Graphs
- Video
- Pod casts and other audio
- Links to informational websites
- Pictures
- Conditional text
- Anything that can be done on the web!
- Organize content, use data to trigger when to present. Examples
- Display sleep hygiene materials (e.g., video, websites, podcasts, local treatmetn providers, etc.) if sleep problems reported)
- Summarize and visualize progress toward key clinical goals across one or more visits
- Automate data-driven calculations (e.g., change in BMI over time, raw score to T-scores)
- Track and list medications
- Examples
- Subject Reports
- A graphical chart of progress toward clinical goals over time.
- Prognostic chart based on the subject's specific clinical characteristics (e.g., a tumor's T, N, M status).
- Patient driven requests for information and/or educational materials (e.g., video on upcoming procedure, list of support groups in the area, referral resources, disease overview pdf file, prognostic chart, table of links to recommended websites, etc.).
- Report of participation points (e.g., provide total points score based on number of forms completed and the points, in turn, can be redeemed for items of interest).
- Study Event Reports
- A report summarizing all adverse events, color-coded with open AEs in red.
- A report showing all safety-monitoring variables or clinical intervention triggers (e.g., depression level over time)
- A study visit invoice documenting and summizing all related charges.
- What To Consider
- What data-driven information would be helpful for key stakeholders (i.e., research staff, clinicians, subjects)?
- What information would help clinical decision making, how is it best conveyed (e.g., graphs,tables, key findings, etc.) and what needs to be documented?
- Automated clinic notes and reports
- What research reports need to be submitted (e.g., adverse event report)
- What data and educational materials would be useful for subjects (e.g., a disease overview and recommendations, display of clinical data over time)?
- What data-driven clinical events will trigger specific information (e.g., if BMI over 25, show video on weight management)?
- How can the report content be customized to fit the specific needs of a subject and provide the greatest incentive for participation?
- Study Event Reports
- What study events will require reporting?
- What template forms need to be completed?
- Adverse Events
- What are the standardized AE and SAE reporting requirements?
- What is the best way to summarize AEs and monitor these status?
- What's the best way to visualize safety-related data?
- What are the safety monitoring variables and clinical intervention cut-points?